Sub-topic: Alloys
Question to ponder: What are alloys?
- A mixture of two or more elements where at least one of the elements is a metal . [E.g. Iron & Carbon = Steel]
- The properties of metal depends on thestructure of the metalitself.
Question to ponder: Since pure metals are soft, why does most metal products around us are harder and stronger?
- Metals can be made harder and stronger by alloying them with other elements.
- The added elements are usually metals. sometimes a non-metal such as carbon is also added. E.g. Steel (made up of iron and carbon).
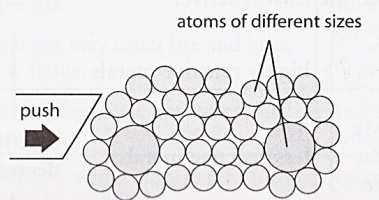
What happens when force is applied to an alloy?
|
Comparison of the properties of metals and alloys:
Pure Metal
|
Alloy
|
Uses of Alloys:
- Brass (Copper and Zinc) : Electric Plug
- Bronze (Copper and Tin): Propellers
- Duralumin (Aluminium and Copper) : Aircraft body
- Pewter (Lead, Tin, some Antimony): Souvenirs
- Solder (Tin and Lead): Joining electrical connections
- Stainless steel (Iron, Chromium / Nickel): Cutlery
The links to download the notes:
PDF VersionNote: You will need a PDF reader in order to read/open this file
[Mirror (via MediaFire): Notes]
For the mirror links, you will need a password to enable you to download these files. Check the password in the forum, under "News" section.Note: I'll activate the direct links later (if I have the time). In the mean time, please use the mirror links for downloading the notes a.k.a MediaFire.
|
Ms Word VersionNote: This document is compatible only with the latest version of Word - 2010 version. Download the compatibility pack if you have an older version of Word.
[Mirror (via MediaFire): Notes 2010]
Note: This note is compatible with older version of Ms Word.
[Mirror (via MediaFire): Notes 2003/2007]
|