Sub-topic: (12.1) Metals and its general physical properties
Metals
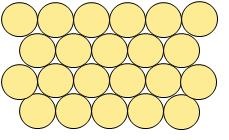
- Most metals are in solid form (except for mercury). Therefore, metal atoms are packed together closely and in an orderly manner.
- You can see the arrangement of pure metals (means that only one element is present i.e. consist of one, e.g. Aluminium, Potassium, Magnesium etc) below:
Look at the arrangement of atoms at the picture!Notice how the atoms are close to each other and that there are little spaces available between atoms. This is how the atoms in pure metal are arranged. The atoms form a strong metallic bond with each other.
This arrangement of atoms of metals explained two of its physical properties: |
(1) Ductile & (2) Malleable
Ductile - Can be stretched into wires.
Malleable - Can be hammered and beaten into shapes.
It is important to note that, although metal atoms form strong metallic bond with each other, this does not mean that they are rigid. Then can still move when force is applied. They move in layers.
Malleable - Can be hammered and beaten into shapes.
It is important to note that, although metal atoms form strong metallic bond with each other, this does not mean that they are rigid. Then can still move when force is applied. They move in layers.
Structure of metal
- In order to explain more about the physical properties of metal, we need to know more than just a simple particle model of atoms; that they are all lining up in close packed rows and layers.
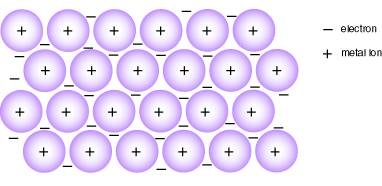
- In fact, metal have the giant metallic lattice structure.
- Remember! The crystal lattice is consist of ions NOT atoms.
- Metal ions easily lose their electrons (E.g. K+ lose one electron easily), causing the ions to be surrounded by electrons.
|
These 'free' or 'delocalised' electrons are mobile. It means they roam freely among the positive metal ions. There is a strong force of attraction between the mobile electrons and the positive ions, forming a strong metallic bond. Electrons are 'negatively' charged.
|
There are 5 physical properties that can be explained by these structures.
(3) High density
(5) Good conductors of electricity and heat
|
(4) High melting and boiling point
(6) Lustrous
|
(7) Sonorous
- Sonorous refers to the ability of the metal to produce sound when struck. How can this be? Again, this is due to the mobile electrons. When struck, the 'sea' of electrons move easily - allowing the incoming energy to be easily propagated in the form of a wave. [i.e. the metal resonate] However, not all metals have this property. Metals need to have a low density in order to do this.
The links to download the notes:
PDF VersionNote: You will need a PDF reader in order to read/open this file
[Mirror (via MediaFire): Notes]
For the mirror links, you will need a password to enable you to download these files. Check the password in the forum, under "News" section.Note: I'll activate the direct links later (if I have the time). In the mean time, please use the mirror links for downloading the notes, a.k.a. MediaFire.
|
Ms Word VersionNote: This document is compatible only with the latest version of Word - 2010 version. Download the compatibility pack if you have an older version of Word.
[Mirror (via MediaFire): Notes 2010]
Note: This note is compatible with older version of Ms Word.
[Mirror (via MediaFire): Notes 2003/2007]
|