TOPIC 2: DIFFUSION, OSMOSIS & ACTIVE TRANSPORT
SUB-TOPIC: (2.2) OSMOSIS
Osmosis is the movement of water molecules from a solution of higher water potential to a solution of lower water potential , through a partially permeable membrane.
Water moves DOWN the water potential gradient .
In comparing two solutions with differentsolute concentrations:
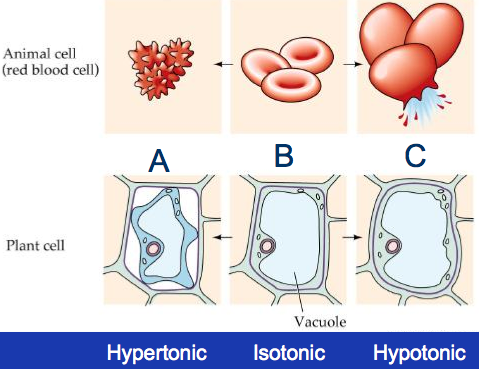
• The solution with higher solute concentration (=low water concentration ) is said to be hypertonic (‘hyper’:more)
• The solution with a lower solute concentration (=high water concentration ) is hypotonic (‘hypo’: less)
• Solutions in which the concentration of solute are equal (=equal water concentrations ) are said to be isotonic (‘iso’: equal)
Water moves DOWN the water potential gradient .
- Osmosis is a special case of diffusion - it only involves water molecules and NOT the solute molecules .
- This process does NOT require energy, hence, called as "PASSIVE TRANSPORT". This is because the movement of molecules are from HIGH to LOW. This process is easy (Imagine yourself walking down a cliff - it's easy compared to climbing up, right?)
- Both animal and plant cell behaves DIFFERENTLY in different solution - mainly due to the present of cell wall in the plant cell.
In comparing two solutions with differentsolute concentrations:
• The solution with higher solute concentration (=low water concentration ) is said to be hypertonic (‘hyper’:more)
• The solution with a lower solute concentration (=high water concentration ) is hypotonic (‘hypo’: less)
• Solutions in which the concentration of solute are equal (=equal water concentrations ) are said to be isotonic (‘iso’: equal)
The Importance of Osmosis
If blood cells (or other cells) are placed in contact with hypertonic solution, the cells will lose water and shrink . Further movement of water molecules to outside of the cell will cause the cell to shriveled, with little spikes appear on the cell surface membrane (crenation). If the solution is hypotonic (or if pure solvent is used) the cells will swell ; the osmotic pressure that is developed may even be great enough to rupture the cell membrane (hemolysis). Saltwater from the ocean is hypertonic to the cells of the human body; the drinking of ocean water dehydrates body tissues instead of quenching thirst.
As for plants, osmosis is important in maintaining turgidity of plant cells. Turgor plays an important role in maintaining the shape of soft tissues in plants. Most leaves and young stems, especially those of herbaceous and non-woody plants (the stems are usually not hard), are able to remain firm and erect because of the turgor pressure within their cells.
So, when there is a high rate of evaporation of water from the cells (basically, the plant lose its water), they lose their turgidity and the plant wilts. Turgor also plays an important part in opening and closing the stomata - guard cells are involved. You will learn more about this in Topic 4.
As for plants, osmosis is important in maintaining turgidity of plant cells. Turgor plays an important role in maintaining the shape of soft tissues in plants. Most leaves and young stems, especially those of herbaceous and non-woody plants (the stems are usually not hard), are able to remain firm and erect because of the turgor pressure within their cells.
So, when there is a high rate of evaporation of water from the cells (basically, the plant lose its water), they lose their turgidity and the plant wilts. Turgor also plays an important part in opening and closing the stomata - guard cells are involved. You will learn more about this in Topic 4.
Preservation of food using salt or sugar
Food is soaked in a concentrated salt solution, so that all the water molecules in the food are drawn out by osmosis.
The food becomes dry causing the bacteria and fungus cannot survive. |
What you can do! [Research]
1. What is reverse osmosis?
2. What are the sources of drinking water in our country? 3. How much water is consumed in our country per day? Note: This is optional. You can do this if you want to. Do this and pass it up to me, and I'll give you extra marks for your assignments :) |
TcNMA <3
The links to download the notes:
PDF versionNote: You will need a PDF reader in order to read/open this file
[Mirror (via MediaFire): Notes]
Ms. Mai's Notes:[Mirror (via MediaFire): Notes]
[No password is required for this note]
For the mirror links, you will need a password to enable you to download these files. Check the password in the forum, under "News" section.Note: The direct link for the PDF Version is already up! You can now directly download the PDF file via this page without having to go to MediaFire! As for the Ms Word Version, I'll activate the direct links after 6 days. In the mean time, please use the mirror links for downloading the notes.
IMPORTANT ANNOUNCEMENT: The direct links have been updated! You can now directly download documents from this website without having to go to MediaFire.
|
Ms Word versionNote: This document is compatible only with the latest version of Word - 2010 version. Download the compatibility pack if you have an older version of Word.
[Mirror (via MediaFire): Notes 2010]
Note: This document is compatible with older versions of Word.
[Mirror (via MediaFire): Notes 2003/2007]
|
||||||||||||||||||